Aging is a fact of life for most, if not all, complex multicellular animals. Aging means simply the regenerative processes work less and less effectively as time passes. We see the signs of it everywhere in our bodies: sagging skin, diminished exercise capacity, increased susceptibility to disease, hair loss, aching bodies, and joints. Eventually we die from a side effect of this degeneration if we do not die from genetic flaw, accident, or mishap earlier. There are some small multicellular organisms, planaria for example, that do not seem to age. Some also argue a few larger organisms, such as some mollusks, crustaceans, and sharks, for which we can find examples of apparently incredibly old organisms with little sign of aging, may not age. We may eventually discover these more complex organisms do slowly age or that they have some secret in their genes that allow them potentially to regenerate and live forever. For most part, however, complex animals age and we do not understand exactly how and why.
Theories abound but all of them seem to have something missing or something that cannot be accounted for. Josh Mitteldorf (who I criticized recently for some of his COVID-19 views) in Cracking the Aging Code has a good summary of main theories and their issues. Certainly, the theory about the body just wearing out does not seem to work out because it cannot account for why maximum lifespans vary from one species to another. Brown rats and mice max out at around four years of age. Parrots and humans can live to around a hundred years or more. There is not any reason rats and mice, kept in laboratory conditions, would suffer more wear and tear than humans. Genetics seems to play a key role since species have been shown to increase their lifespan when natural selection pressures change. Opossums living on islands without heavy threat of predation gradually changed to live 25-50% more than the same populations living on the mainland. The fact that some species, such as species of salmon, seem to age little if at all until a certain age then decline rapidly, usually after reproduction, suggest that aging, as Mitteldorf believes, is a genetic program.
Still exactly how the program kicks in, how it works, and whether there are things that can be done to modify significantly the program are open questions.
The quest for how and why has looked at glands, hormones, mitochondria, and telomeres. In the early 20th century, Élie Metchnikoff, Russian zoologist who gained fame as an immunologist, suggested in The Prolongation of Life: Optimistic Studies an association between the gut and aging. While his exact theory, that aging is caused by toxic bacteria in the gut, is not correct (although toxic bacteria could be an influence on the aging process), he may have been the first modern scientist to suggest that the gut was directly involved in aging. He also thought, by the way, that lactic acid and consumption of yogurt could prolong life.
While there are today an increasing number of studies showing the effect of aging on the gut and how the gut’s microbiome affects health, to the best of my knowledge no one is proposing the aging program is directly initiated from the gut or from the gut brain axis. While aging certainly affects the entire body, I am speculating there may be a direct connection between the gut and aging.
Let us look at several lines of evidence of connection between the gut, the gut microbiome, and aging.
Caloric restriction extends lifespan.
Calories come through the digestive actions in the gut. Restricting calories while maintaining optimal nutrition has been shown to extend lifespan in rats, mice, fish, flies, worms, and yeast. The earliest evidence of this dates from the 1930s in studies by McCay. Calorie restriction extends the lifespan of C. elegans by 40%. Calorie restriction improves markers for aging in humans but has not yet been shown to extend lifespan. Caloric restriction produces structural changes in the gut microbiome. It is not understood how calorie restriction works, but large number of humans are experimenting with various techniques of caloric restriction in hopes of extending their life. Common techniques include daily lower calorie consumption, every other day fasting or restricted eating, and a technique tested by Valter Longo that involves five days a month of restricted calorie and low protein eating.
Intestinal system and gut microbiome changes with age.
The intestinal immune system is impaired in aging and the elderly show increasing incidence of GI tract infections and disorders. Changes in the microbiome in humans and fruit flies have been shown to correlate with aging. Types of microbes in the gut can be used to measure age and scientists have created a human microbiome clock that can predict age within four years. Age related butyrate production, normally done by healthy microbes in the gut, has been shown to be related to neurogenesis, hypertension, and heart disease.
Alteration of the gut microbiome extends life.
Scientists extended the lifespan of fruit flies by 60% by feeding them probiotics and an Ayurvedic herbal supplement called Triphala. The treatment also protected against chronic diseases associated with aging. Various interventions that improve the gut microbiome also seem to have positive effects on diseases associated with aging. Research at Scripps Research suggests a possible mechanism that might relate to longevity. Scientists developed molecules that can remodel the bacterial population of intestines to a healthier state and this reduces cholesterol levels and strongly inhibits the thickened-artery condition known as atherosclerosis.
Metaformin and rapamycin, drugs suggested as potential life extension agents, may work in part by changes in the gut.
Metaformin has been shown to improve multiple metabolic indicators associated with diabetes and aging across various species. It has been suggested as a life extension drug. One study showed that both, metformin and phenformin decelerate aging in C. elegans in dose-dependent manner and increased lifespan up to 26-36%. Although metaformin modifies various metabolic pathways, the mechanism metformin increases lifespan in C. elegans is by alteration of microbial folate and methionin metabolism. Metaformin has been shown to produce alteration in gut microbiota and to reduces aging-related leaky gut while improving cognitive function by modulating the gut microbiome and other intestinal factors. Rapamycin, another drug with evidence for life extension, may work in part by repairing the leaky gut and apparently does not need microbiota to do that, although it also seems to have effect on microbiota too perhaps indirectly through its effect on intestinal wall cells.
Aerobic exercise changes the gut and extends lifespan.
Aerobic exercise has been shown to have positive effects on various markers for age. A study indicates it may extend life expectancy by 4-8 years. Aerobic exercise also increases the abundance and diversity of beneficial bacteria in the gut.
Sleep cycle disruption accompanies aging and causes changes in the gut.
A recent study has shown that gut microbiome diversity is associated with sleep physiology in humans. The authors write: “Analysis of microbiome composition revealed that within phyla richness of Bacteroidetes and Firmicutes were positively correlated with sleep efficiency, interleukin-6 concentrations and abstract thinking… Our findings initiate linkages between gut microbiome composition, sleep physiology, the immune system and cognition. They may lead to mechanisms to improve sleep through the manipulation of the gut microbiome”. Aging in both humans and fruit flies is associated with changes the gut microbiome.
At this point, we might be tempted to think that Metchnikoff’s idea of aging caused by toxic bacteria might be more right than wrong. The fly in the ointment for the theory is that germ free mice without gut bacteria eventually age and die too. Importantly, the mice do live longer than mice on unrestricted diets, although not longer than mice fed on a restricted diet; however, they suffer permanent neurodevelopmental deficits that may them useless for researching anything else than the relations between the gut and brain. This suggests to me two things. One is that aging cannot be explained simply by toxic bacteria in the gut. Alterations to the gut microbiota might stave off aging but not free ourselves from it. The other is that the gut microbiota has had a long and complicated evolutionary relationship with complex animals that may go back almost to their origins. The microbiota of the gut play an essential role in the development of the brain. This relationship could have had significant impact not only on the brain but also on aging.
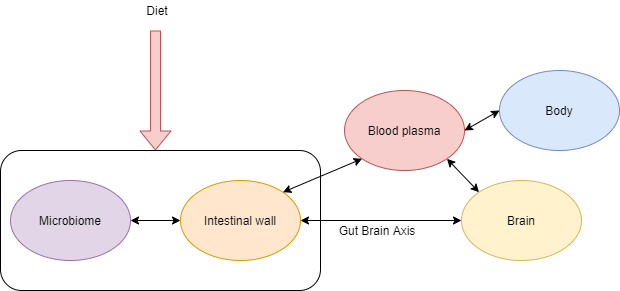
The gut or brain gut axis may be the lowest common denominator of complex animals with brains that age. Some of the earliest ancestors of complex animals, the bilaterians, were living tubes consisting of a digestive tract with entrance at one end and an exit at the other. Near the mouth there was a concentration of neurons that later evolved into a brain. Running down the digestive tract was a neural cord that evolved into a spinal cord. The neurons were there to control the mouth and the digestive process. The digestive system was there to provide fuel for the neurons and the body. Aside from a reproductive system and rudimentary circulatory system, the worm has no other organs.

Basic Bilaterian Structure
What is not normally appreciated, but is clearly shown in the above diagram, is that technically speaking the inside of the intestine is outside of the body. The alimentary canal is open at both ends so food, waste, and billions of bacterial cells of the gut are outside the body. The intestinal wall functions to contain the digestive process, to admit nutrients and products of digestion into the body, and to bar harmful bacteria and their toxins from the body. That harmful bacteria is going to get into the body through the normal process of eating is inevitable. The evolution of a symbiotic relationship between good bacteria and the intestine not only aids the body with digestion by enhancing the digestive process, including digesting material that could not otherwise be digested, but also it serves to contain the harmful bacteria by reducing its ability to multiply.
The innermost part of the intestines is the epithelium. It is where most of the secretion and absorption processes associated with digestion occur. It both affects the microbiota with its secretions but also is affected by the microbiota. Surrounding it are several layers. One of which is the enteric nervous system that governs the gastrointestinal tract. It can act independently from the rest of the nervous system and is sometimes called the second brain. Familiar neurotransmitters like serotonin and dopamine are present in the gut. It has 500 million neurons, more neurons than the spinal cord.
Gut-brain Axis
Today we are discovering close relationships between the digestive system – the gut – and the brain. The gut contains the largest number of neurons in the body outside of the brain. Our understandings of this connection are relatively recent, and much study is still underway, but the term “gut-brain axis” is increasingly popping up in the literature.
The gut-brain axis (GBA) consists of bidirectional communication between the central and the enteric nervous system, linking emotional and cognitive centers of the brain with peripheral intestinal functions. Recent advances in research have described the importance of gut microbiota in influencing these interactions. This interaction between microbiota and GBA appears to be bidirectional, namely through signaling from gut-microbiota to brain and from brain to gut-microbiota by means of neural, endocrine, immune, and humoral links.
Aging may be a genetic program, as Mitteldorf thinks, that is built into almost every cell in the human body. If that is the case, there will probably little we can do fundamentally about aging without making changes in the human genome. Whether that will be possible without a significant alteration in the human species is probably an open question. To live essentially forever, we may need to become something other than human.
Even if it is the case that aging is built into every cell, it still might be that the way the aging program implements itself is primarily through the gut. In other words, something in the gut or the gut-brain axis may be the primary limiting factor for life extension. The most logical candidate in my view for the weak link would be the epithelial layer of the gut, but it could be and likely is a variety of interactions and not one single thing in the gut-brain axis. How exactly all of the signalling works is hazy but it seems likely there are a lot of different feedback processes at work.
Leaky gut syndrome, a dysfunction in the intestinal barrier, is a key marker for a variety of diseases, many of which are associated with aging, and is directly tied to dysfunction of the epithelium.
From another fruit fly study:
Similarly, intestinal barrier dysfunction is more accurate than chronological age in identifying individual flies with systemic metabolic defects previously linked to aging, including impaired insulin/insulin-like growth factor signaling, as evidenced by a reduction in Akt activation and upregulation of dFOXO target genes. Thus, the age-dependent loss of intestinal integrity is associated with altered metabolic and immune signaling and, critically, is a harbinger of death.
That the gut itself would be the limiting factor for life would not conflict with the observation that lifespan appears to be somewhat evolutionarily determined, as we saw in the opossum example. In fact, it would even be evolutionarily logical in a sense, not that we can expect evolution to be always logical. For one thing, it would require only slight alterations in the genetic or epigenetic code that affected the epithelial cells (for example) to raise or lower the lifespan of a species. For another thing, the gut itself is common to all complex animals so the lifespan mechanism would be present in almost all species. Planaria, which some believe to be potentially immortal, have an unusual digestive system and apparently can regenerate any part of their body which would include the cells of the gut.
Whether the gut or the gut-brain axis is key to longevity or not, increasingly research is indicating its significance in a wide range of disease conditions. If the origin of most of the problems in aging is a gradual dysfunction of the epithelial layer of the gut, probiotics may mitigate the problem but eventually the layer ceases to function correctly, possibly because of some limit in the epithelial stem cells. In the meantime, there is a lot we can act on from what we do know. The recommendations, of course, are not all that surprising. Get more exercise. Eat better and get enough fiber. Incorporate some fermented foods into your diet – yogurt, sauerkraut, kimchi, miso, and such. Consider taking some supplements: probiotics, prebiotics, Triphala, and butyrate. You won’t live forever but you may live a little longer and little healthier.

I am going to have to read this again. I didn’t know your interests extended into this. :o)
LikeLiked by 1 person
Don’t forget to click on the links too. 🙂
Actually I really did spend a lot of time getting the links into this post. And I must have left out about 15 or 20 I could have put into this.
It’s incredible how much information is out there on this topic.
LikeLike
Interesting survey James!
In the end it seems like a probability game. I had an uncle who did everything right, ran several miles a day, ate healthy, didn’t smoke or drink, and died of cancer in his 50s. My own Dad, who smoked like a chimney, didn’t seriously exercise or eat particularly well, and never went to the doctor, lived to 81. We can do things to increase the probability of longevity, but that’s about it.
But it seems like there’s a cost / benefit ratio to consider here. We can do some basic things and get most of the longevity benefits, but beyond a certain point you’re exerting a lot of effort for marginal benefit. It’s the old 80 / 20 rule; you get 80% of the benefits for 20% of the effort. And entropy can only be held off so long.
LikeLiked by 1 person
We do need to distinguish aging from genetic defects and deaths from other causes like infectious diseases, cancer. Aging can increase the probability of cancer but its causes may be many and could include viral agents, chemical or radiation exposure, genetic propensity, or probably some complex interaction of various things.
But you’re right about the 80/20 rule and probably excesses even in what we might think to be healthy habits can be harmful. I think there is an association of asthma with growing up in a too clean, too germ-free environment. Roy Walford, one of the leading proponents of calorie restriction, actually died relatively young. Whether the restricted diet he followed was itself part of the issue is hard to say. There is also the hormesis phenomenon where a little bit of poison or radon radiation can actually be good for you. In a way, vaccines work by giving your body exposure to a mild case of disease.
LikeLiked by 1 person
One other note on entropy. Before aging sets in and even afterward, the body has the ability of renew and recreate itself. Species like planaria seemingly can maintain this ability forever. There is no entropic reason that a living organism must break down. Living organisms are programmed to do so. I do agree with Mitteldorf on this point and he devotes a few pages to the argument in his book.
LikeLiked by 1 person
I’m not well read in this area, so this might be a very basic question. Are there any more complex species other than ones like planaria that don’t age? I remember once reading something about some birds possibly not aging, but I can’t recall where.
If evolution is for the genes, then it would make sense that individual organisms are programmed to age out. One of the issues I also recall reading is that humans, due to the grandmother effect, are already on the extreme end of that in terms of longevity. So, according to this article from a few years ago, a lot of things that extend life in other animals won’t work on us, because our expected life spans are already longer than most species our size.
LikeLiked by 1 person
Actually mentioned those in the first paragraph.
“Some also argue a few larger organisms, such as some mollusks, crustaceans, and sharks, for which we can find examples of apparently incredibly old organisms with little sign of aging, may not age. We may eventually discover these more complex organisms do slowly age or that they have some secret in their genes that allow them potentially to regenerate and live forever. ”
I think the Greenland shark is certainly a potential candidate since some of them have been found that might have been around for 500 years. I don’t recall anything about birds. Parrots can live to a 100 or so, but I think they do age.
In that light, sharks do have an interesting digestive track with something called a spiral valve and I think the food is almost completely digested when it reaches there. It doesn’t look like it works like intestines in other species. I was looking into that while working on this piece but haven’t delved too deeply into it.
I also looked into Pacific salmon which age very rapidly after spawning and found an article that shows significant changes in the intestine: “The tall epithelial cells of the sea salmon tend to become low columnar and in certain instances have shrunken to an almost cuboidal shape.” Also, number of epithelial cells are reduced in number.
Of course, it is possible these changes in the salmon and changes in fruit flies and humans are caused by aging rather than playing an active role in causing aging. My rationale for the gut being causative is primarily based on two things:
1- Aging is common to many animal species from worms to humans so there may be a common mechanism.
2- The digestive track is one of the few things that is common to worms and humans.
Although after posting this, it occurred to me that obviously reproductive organs are common. Since aging particularly in some species (Pacific salmon) seems closely tied to reproduction, it could be that the aging is tied to the triad of gut, brain, and reproductive organs. The problem with that is humans can have reproductive organs removed without apparently aging quickly so I would think in humans the reproductive system would play a minor role.
Regarding human potential I don’t think we can be sure. I keep thinking of the opossum extending its lifespan up to 50% through natural processes. Some evolutionary process may be working on humans too, although certainly most of our life extension has occurred with medicine and sanitation. But what is possible is hard to say. There are some recent posts on Mitteldorf’s site about plasma replacement providing a rejuvenative effect. Reading those in part was got me off on this track because it seemed to me that, if there are factors in blood plasma that cause or retard aging, the factors would likely come from the gut. Who knows what might be possible in the future with stem cell therapy. Let’s say epithelial cells are a limiting factor. I could easily imagine some sort of stem cell therapy could potentially regenerate epithelial cells and we might gain another fifty, probably healthy, years or so.
LikeLiked by 1 person